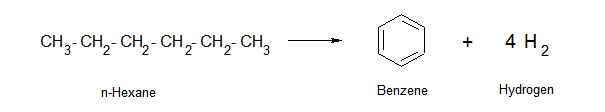RL Blogs
By Optimization Specialist Robert
May 01, 2017Understanding the value of benzene optimization in gasoline production can yield high value. |
||
| Have you ever wondered what value you can extract by improving gasoline blend performance related to benzene specifications?
With reduced benzene specifications in North America driven by MSAT II regulations, many technology firms have sought to sell new processes to manage gasoline benzene. While many refineries required technological solutions to meet compliance, there are many who do not. Furthermore, for those who have achieved lower benzene specifications, they may be over-complying, thus giving away margin.
For refineries without chemical complexes, benzene is primarily formed in the reformer unit. Without extraction equipment, refiners can either blend benzene directly into finished gasoline, or re-saturate these compounds into a less toxic naphtha derivative. Either way, managing benzene has a cost, and a refiner’s ability to optimize around this cost can be a big winner or a big loser.
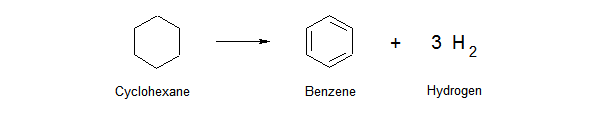
Let’s first cover some basics on how benzene can be formed. The easiest mechanism to create benzene is to extract hydrogen from a cyclohexane molecule in a refinery reformer unit. An abundant amount of hydrogen is formed in this reaction, along with a benzene molecule.
As another example of benzene creation, normal hexane can undergo ring formation and release hydrogen to form a benzene molecule.
When it comes to refinery benzene management, there are two questions that come to mind:
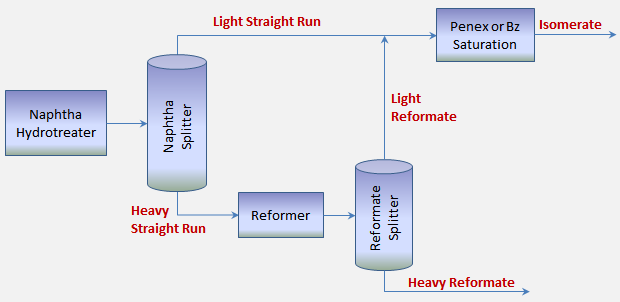
In addressing the question of minimizing benzene creation, we will focus our attention to Naphtha Splitter operations. In most refineries, engineers and operators have the ability to manage the quality of feed going to the reformer. Strategies exist to manage the Light Straight Run (LSR) and Heavy Straight Run (HSR) cutpoint.
While there are many more naphtha precursors that can create benzene than discussed above, the key is understanding the main interface of C6 isomers where the efficiency of benzene production falls off a cliff. For naphtha reformers, this interface occurs between Methylcyclopentane (MCP) and Cyclohexane (CH).
The reaction speed of forming benzene from Cyclohexane is rather quick, while that of Methylcyclopentane is not. Therefore, fractionation strategy in naphtha splitters upstream of a reformer should focus on minimizing MCP in the Heavy Straight Run while maximizing the CH content.
Refiners with gas chromatograph measurement capabilities can achieve this fairly easily. For refiners without GC capabilities, engineers can simulate a distillation temperature profile based on feed properties and fractionation column details.
The real question is what sets the LSR/HSR cutpoint? The answer should depend on the type of equipment downstream of the naphtha splitter. If the refinery does not have some form of benzene destruction technology (i.e. Bensat or Penex), then the driver of the LSR/HSR cutpoint should be fully devoted to controlling the production of benzene. Refiners with this configuration should focus on managing the benzene precursor content in the reformer feed. These refiners should cut the LSR and HSR so that the ratio of Methylcyclopentane and Cyclohexane in the Heavy Straight Run yield the target Benzene concentration in the reformate product blended into gasoline. This is required since these refiners have no way of removing benzene from the refinery once it is created.
For refineries that have some form of benzene destruction technology, the question of benzene optimization becomes less clear. The reason for this ambiguity is because yield performance of the downstream units may influence the economics of benzene yield. For example, some refiners may find it more economic to minimize the production of benzene as the primary strategy. Other refineries may rely on destroying benzene after it is produced.
When removing benzene, refiners should recognize that all of the hydrogen created during the benzene formation process must be consumed when destroying it. Benzene saturation is a quick process in the presence of hydrogen and catalyst.
The table below compares some pros and cons of each approach:
As you can see, there are several pros and cons with each approach above. While finding the optimal solution is important, the more critical learning is that refiners recognize that various solutions exist. Having this depth of understanding provides the comfort that gasoline benzene can be properly managed, thus refiners should target this as a key focal point for optimization.
So what is the value of improving gasoline benzene giveaway performance? It can be summed up into 3 key drivers:
All of the above are worth significant margins to refiners today given the many constraints that they face. Like many other product specifications that exist, gasoline benzene is not one that is often directly managed by the gasoline blender. As a result, a significant amount of giveaway occurs and this costs refiners significantly.
The ability for operations, engineers, and planning analysts to identify the opportunity and close the gap will deliver strong results. In conclusion, your refinery should have no excuse if benzene giveaway exists in the gasoline pool. | ||
|
|